Many of us wonder what a class III medical device means as we hear about it more often these days. Well, simply put, class III medical devices are those equipment that have a higher risk to users and patients. To be more precise these are devices planted inside the body that actively support and sustain lives.
The Food and Drug Administration (FDA) regulates medical devices and has classified them into three groups. As mentioned earlier, Class III devices have the highest risk profile when compared to Class I and II. We will talk more about the medical device classification and its wider implications in the sections below.
Things That Make A Device Class III
It is always good to know about the factors that make a device qualified for class III status. Check out the following.
- Devices with known risks and unable to control
- Devices with risk-benefit profiles that are not favorable
- Equipment that requires a full manufacturing review of information
- Devices that have received a pre-marketing review regarding a change in its effectiveness and safety
- Combination products with a primary device model of action starting the drug constituent is effective and safe
- A piece of high-risk equipment suitable for sustaining human life and holds substantial importance in preventing health impairment

The list of class III medical devices
Here, we will list the medical devices that belong to the class III medical devices. See below for details.
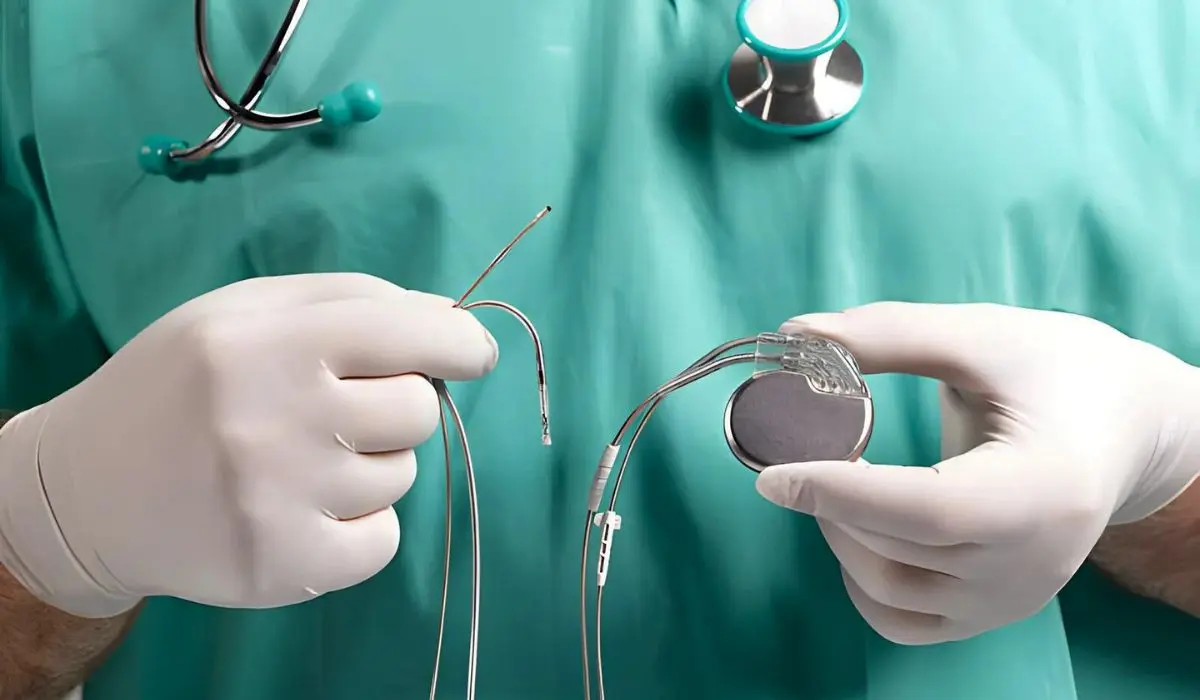
♦ Pacemakers
A pacemaker is a small device powered by a battery to prevent the heart from beating too slowly. It is normally placed under the skin near the collarbone via surgery. There are three different types of pacemakers such as single chamber pacemakers, dual-chamber pacemakers, and biventricular pacemakers.
♦ Implanted Prosthetics
Medical implants are generally devices that are placed inside or on the surface of the body. Many types of prosthetic devices offer assistance to regain their mobility. The prominent among them are transracial, transhumeral, transtibial, and transfemoral to name a few.
♦ Cochlear Implants
Cochlear implants belong to the class of small electronic devices that provide a sense of sound to individuals who are deaf or face challenges in hearing.
♦ Defibrillators
The Defibrillators are life support devices that apply an electric charge to restore a normal heartbeat. During instances of cardiac arrest, this device may help it start beating again.
♦ Software defined as a medical device (SaMD)
It is a class of software that uses imaging functions to identify conditions that if missed will pose a greater risk to the patient’s life.
FDA regulatory approval process for class III medical devices
It has to be noted that almost all class III medical devices require premarket approval (PMA) from the FDA before being introduced into the markets. The PMA process is very time-consuming and requires significant data to demonstrate the safety and effectiveness of the device. This is due to the high-risk profile of class III devices.
The FDA will thoroughly review the PMA process and approve the product for entry into the US markets. The process of PMA is well defined in the Title 21 Code of Federal Regulations (CRF) Part 814. Given below are some of the requirements of the PMA.
- Substantial clinical trial data
- A fully documented quality system compliant with design controls as defined in 21 CFR Part 820
- Documented conformance to recognized consensus standards
- Detailed descriptions of each component of the device
- Product samples and/or allowing the FDA to evaluate the device on-site
Post-market compliance for class III medical devices
The manufacturer of class III medical devices and distributors should abide by certain specific requirements once a product gains entry into the markets. These requirements include:
- Mandatory reporting of device issues and adverse events by manufacturers, importers, and device user facilities as defined in the Medical Device Reporting regulation (21 CFR Part 803)
- The implementation of tracking systems to facilitate product recalls as specified in 21 CFR Part 821
- Post-approval studies with the approval of a PMA, Humanitarian Device Exemption (HDE), or Product Development Protocol (PDP).
Also Read:- What Are The 5 Warning Signs Of Heart Disease? Stay Alert!
Conclusion
In this article, we have had an overview of the class III medical devices and the factors that make them qualify for the status. We listed out the devices that belong to the class III device list and also saw the FDA regulatory approval process for class III medical devices.